 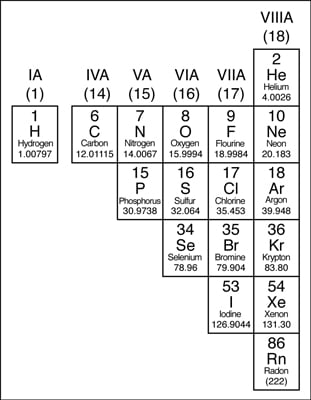
So in practice we're not really combining elements rather atoms of different elements. A selenium speciation machine can measure the amount of selenium present.So elements are different types of atoms which are differentiated by their atomic number (the number of protons). Some semimetals like selenium and arsenic can be toxic. Germanium is heavily used in the semiconductor industry. They act as metals when reacting with halogens, and as non-metals when reacting with alkali metals. Most form several different anions with oxygen that make a variety of salts with metals, although the aqueous chemistry of germanium is more limited. Metalloids generally have chemical properties similar to non-metals. Semi-conductors are capable of conducting electricity better than insulator, but not as well as conductors. 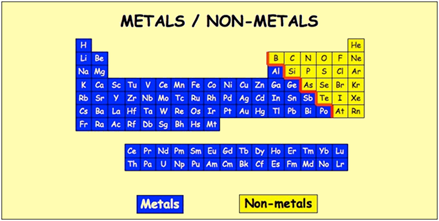
They are brittle, and are typically semi-conductors. Some forms of selenium have an amazing, almost space-like sheen. Metalloids can be shiny or dull, but usually have a metallic luster. Semimetals, also known as metalloids, have properties of both metals and non-metals. Most of non-metals exist in two of the three states of matter at room temperature: gases and solids, except bromine, which exists as a liquid. They tend to have lower melting points than metals. In general, non-metals are brittle, dull, and poor conductors of heat and electricity. Thus, they are electronegative elements with high ionization energies. Nonmetals are elements that form negative ions by gaining electrons during chemical reactions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
